REF. BJS Jan 16 special issue. Wiki on sarcopenia.
We appearantly can, and this is how:
Similarly to the 1996 World Health Organization (WHO) methodology for definitive diagnosis of osteoporosis, which uses DEXA, which uses a cut point of 2 standard deviations below the mean of DEXA results for gender specific healthy young adults to define osteoporosis. In here, we use a measure of lean mass rather than bone mineral density (BMD).
The European society on clinical nutrition and metabolism special interest group on geriatric nutrition had a concerns us definition for sarcopenia:
1) A low muscle mass, >2 standard deviations below that mean measured in young adults (aged 18–39 years in the 3rd NHANES population) of the same sex and ethnic background, and
2) Low gait speed (e.g. a walking speed below 0.8 m/s in the 4-m walking test).
Linda Fried / Johns Hopkins Frailty Criteria Edit
A popular approach to the assessment of geriatric frailty encompasses the assessment of five dimensions that are hypothesized to reflect systems whose impaired regulation underlies the syndrome. These five dimensions are:
unintentional weight loss,
exhaustion,
muscle weakness,
slowness while walking, and
low levels of activity.[1]
Corresponding to these dimensions are five specific criteria indicating adverse functioning, which are implemented using a combination of self-reported and performance-based measures. Those who meet at least three of the criteria are defined as “frail”, while those not matching any of the five criteria are defined as “robust”.
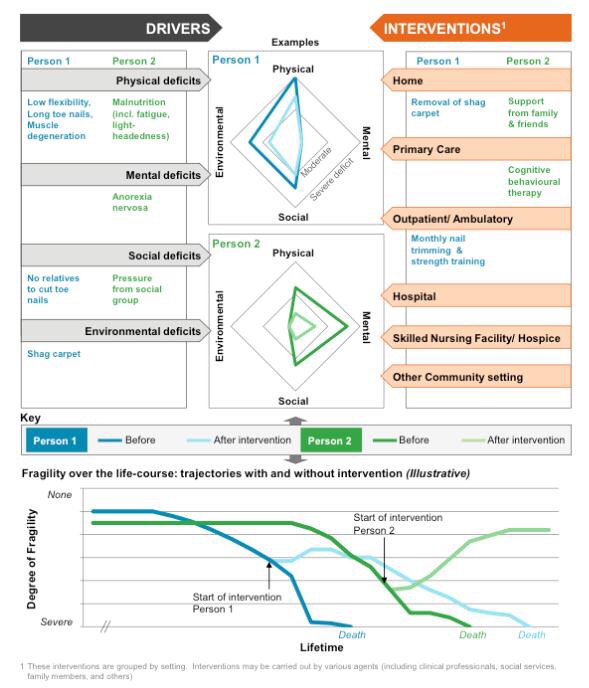
The Bmj (based on research commissioned by National Institute for Health Research (NIHR) under the Collaborations for Leadership in Applied Health Research and Care (CLAHRC) programme for North West London) provided another way of measuring fragility, as in the following figure:
Authors in the BJS JAN 2016 measured the psoas as volume as a proxy for sarcopenia, and showed a significant predictive power to estimate the risk of frailty on patients undergoing major HBP surgery.
Nhs England has produced a document on how to measure frailty and how to provide a better care to elderly population.
